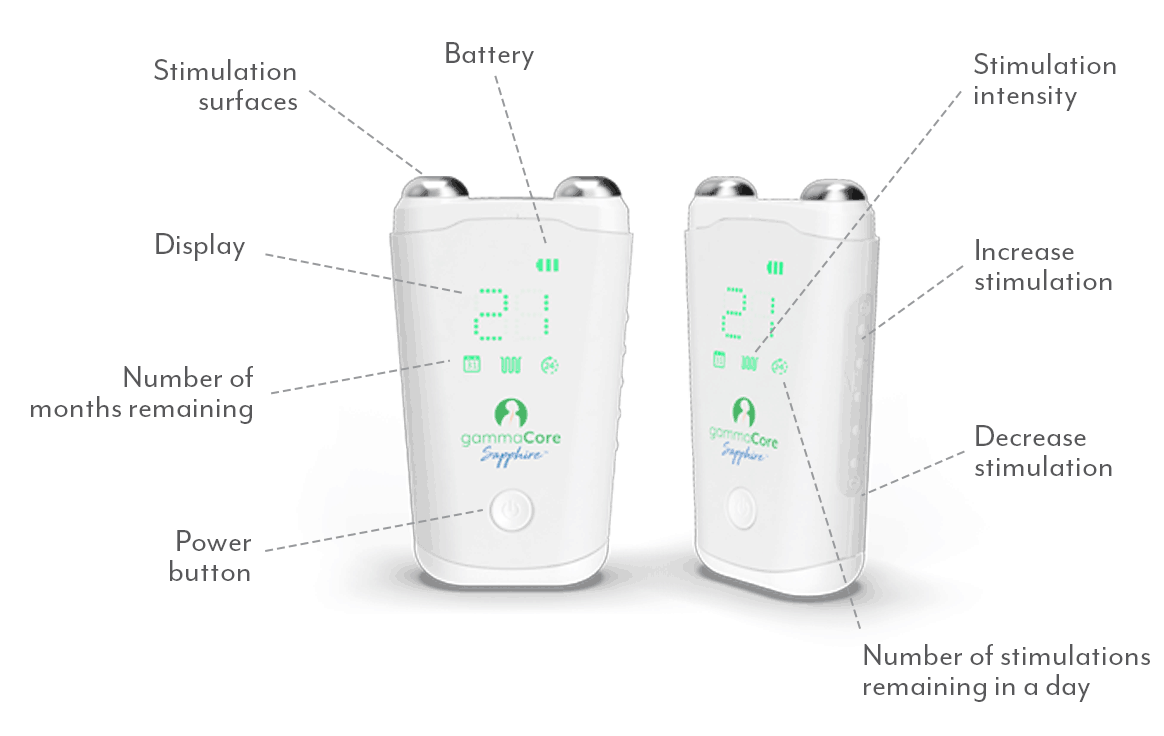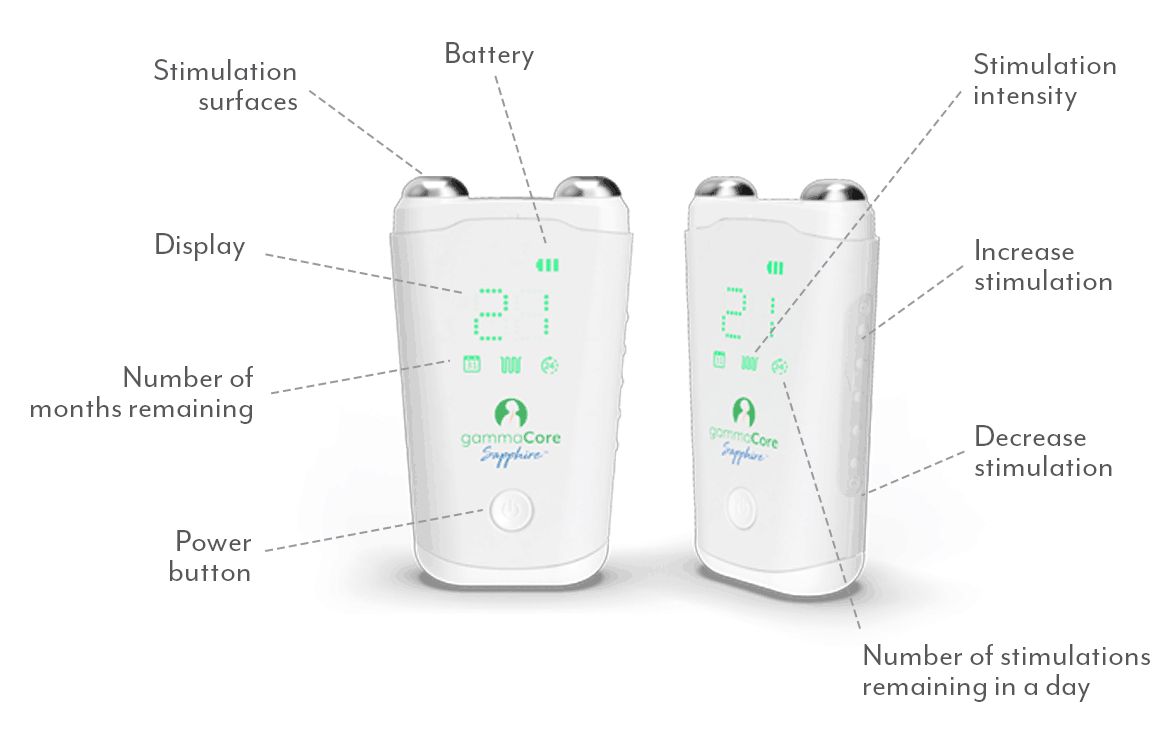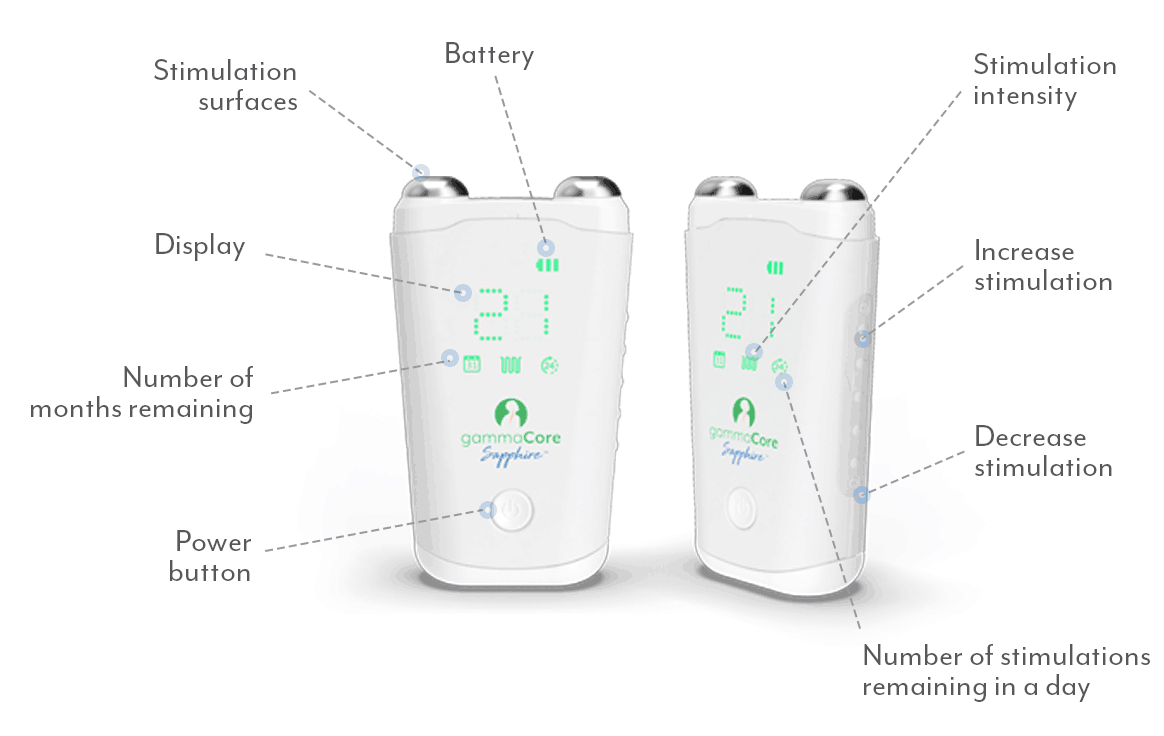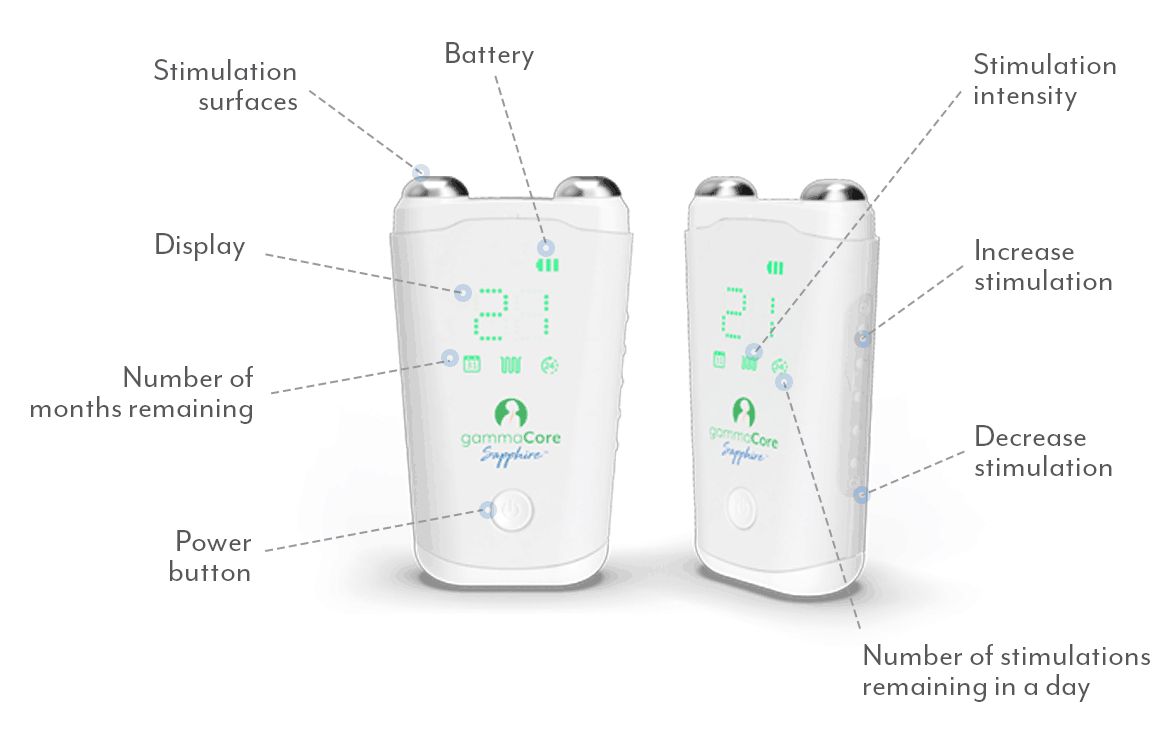
Put the power of non-drug relief in your patients' hands
Consider gammaCore: a non-drug treatment for migraine headache
Migraine pain not only impacts a patient’s body, it impacts their life. Choosing gammaCore (nVNS) will provide your patients with a safe, veratile therapy that can be used both acutely and prevantatively.3-5
Significant Reduction in Migraine Days (mITT)1
A prospective, double-blind, sham-controlled, randomized study evaluated the ability of nVNS to prevent migraine days.
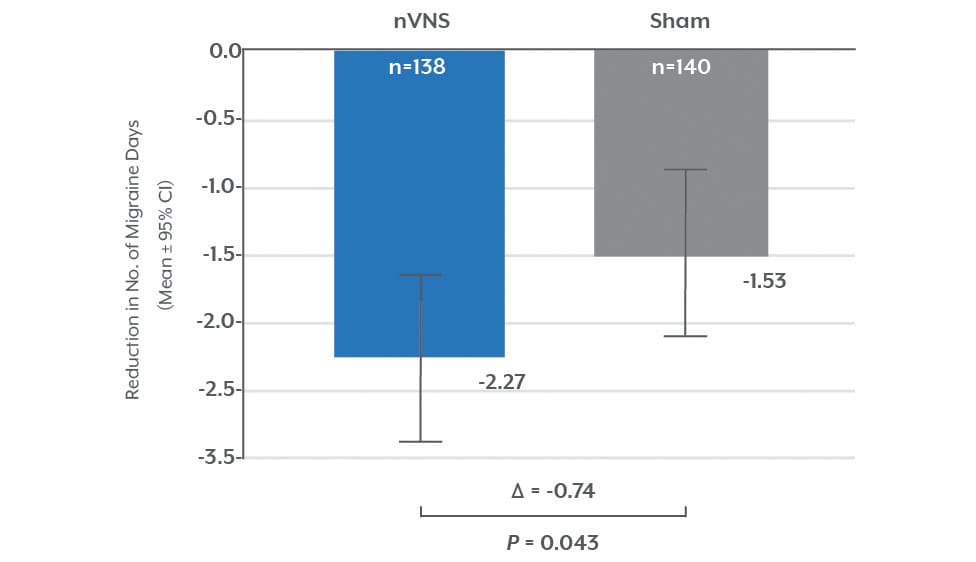
• 25% reduction in migraine days per month from 8 migraine days for patients suffering from episodic migraine.
• Therapeutic gains with nVNS were greater for patients with aura (-2.96 days).
≥50% of Migraines Successfully Treated (mITT)1
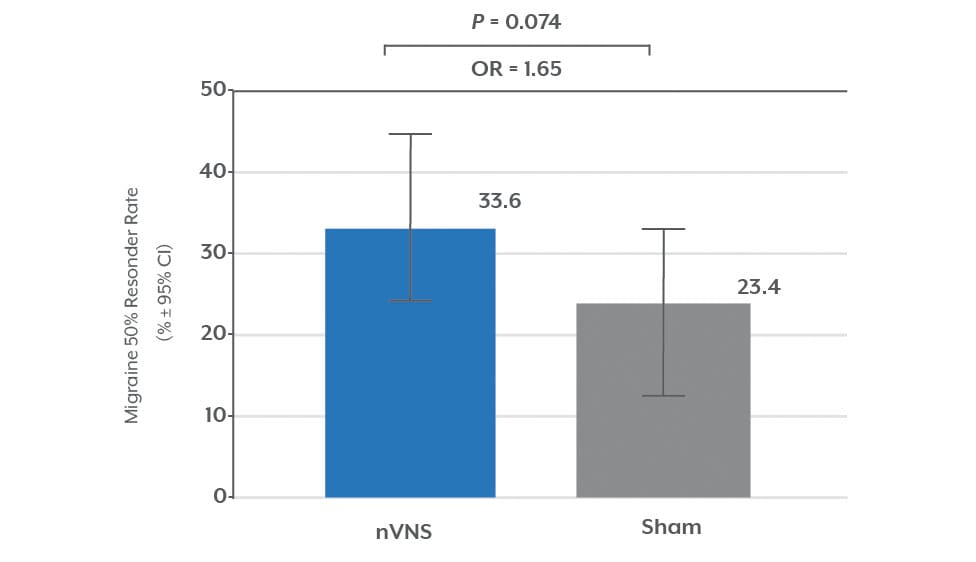
• One in three patients had >50% benefit.
Intent To Treat (ITT) n=332; Modified Intent To Treat (mITT) n=278. Results are presented from the mITT population. Results from the ITT population were not significant.
Continued Use May Enhance Efficacy2
A prospective, double-blind, sham-controlled randomized study evaluated the ability of nVNS to prevent migraine days2
nVNS Treatment Duration
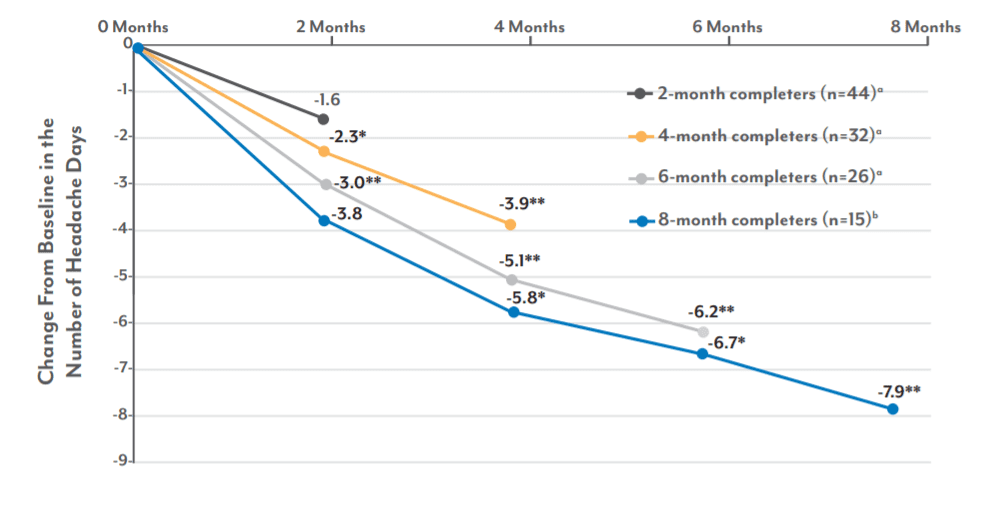
• Mean change from baseline in number of headache days per 28 days through the duration of nVNS treatment for patients with >15 headache days per month.
• Patients adherent to therapy reported continued decreases in headache days.
Clinically proven as an effective treatment for migraine pain3,4
A double-blind, randomized, sham-controlled trial evaluated the safety, efficacy, and tolerability of gammaCore in 243 patients with migraine headache
Study Design
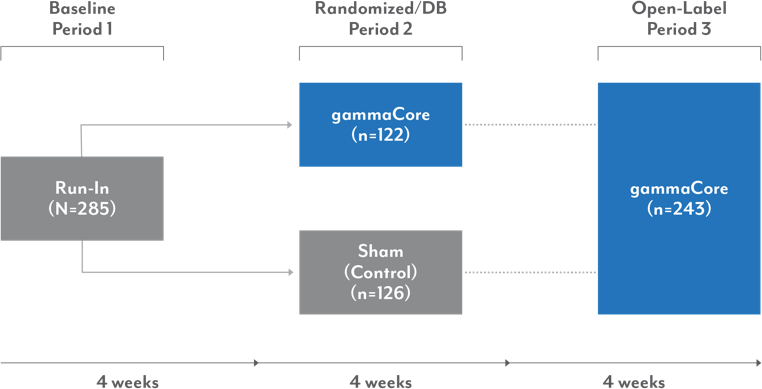
Primary endpoint: Pain-free at 120 minutes; first attack.
Study population: Patients aged 18-75 years, who had been diagnosed with migraine and experienced 3-8 migraine attacks per month and less than 15 headache days a month, were included.
Fast relief from the first treated migraine3-5
gammaCore provided significant relief in as soon as 30 minutes
Percentage of Patients Who Were Pain-Free for the First Migraine
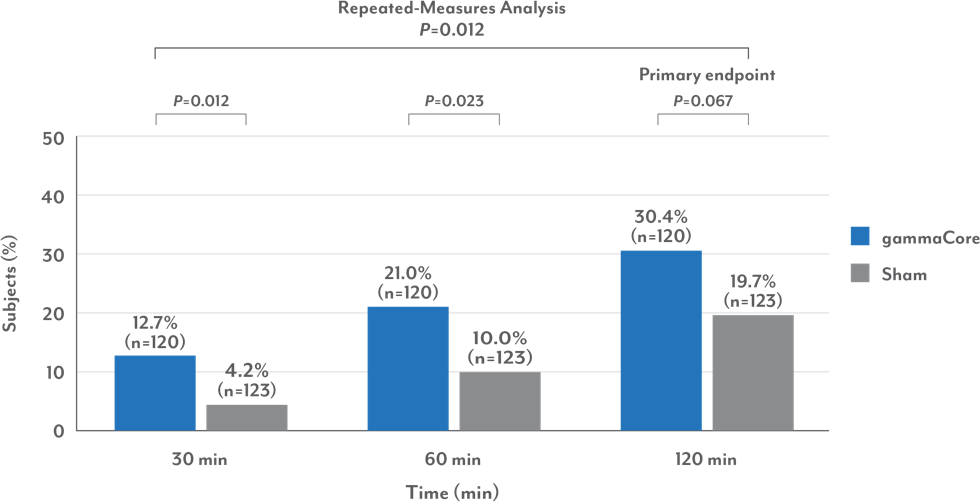
• gammaCore provided improved relief vs sham at 30, 60, and 120 minutes
• 75% of patients who were pain-free at 2 hours were also pain-free at 24 hours
• While more patients experienced relief with gammaCore at 120 minutes (primary endpoint), it was not statistically significant
Reliable relief across multiple migraines3,5
gammaCore provided reliable relief for more patients vs sham
Migraine Patients Successfully Treated ≥50% of Migraines
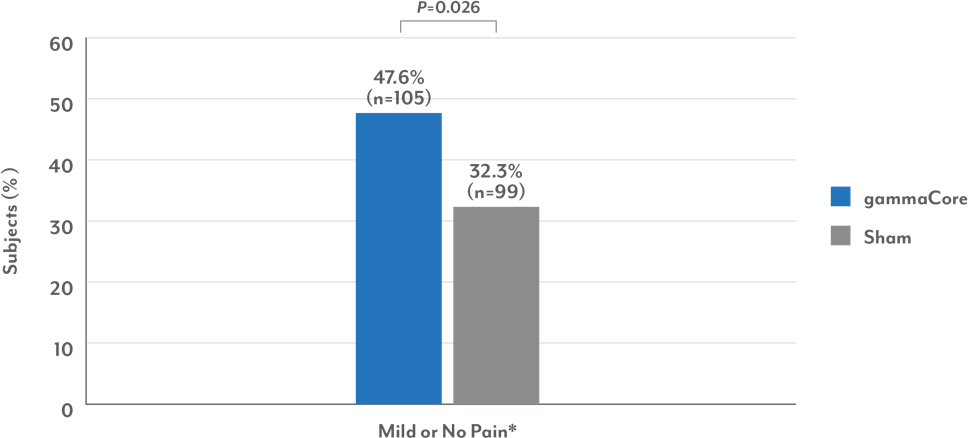
*Patients with mild pain at both baseline and 120 minutes were not considered responders.
• Almost half of the patients treated with gammaCore reported mild or no pain at 120 minutes for more than 50% of all treated headaches
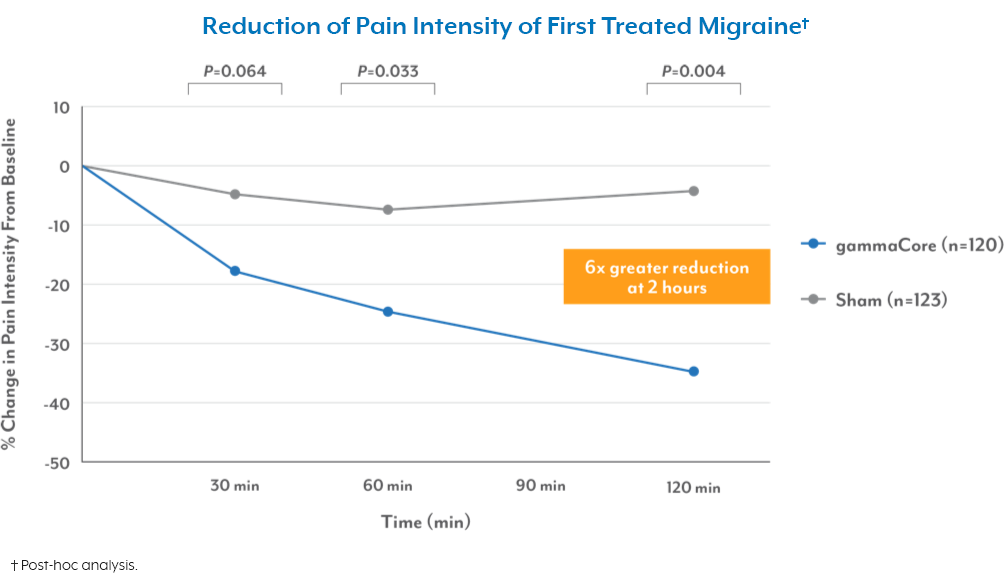
Reduction in pain intensity for first migraine3-5
gammaCore significantly decreased pain intensity vs sham
Reduction of Pain Intensity of First Treated Migraine†
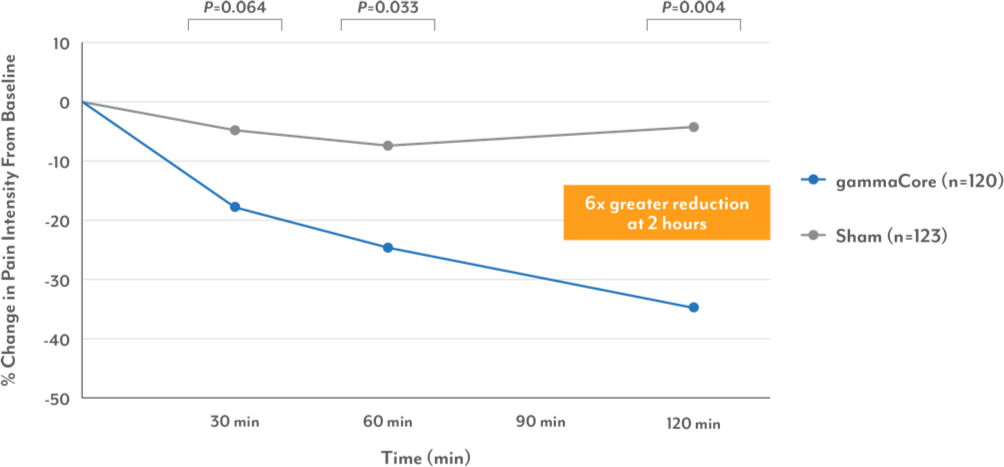
†Post-hoc analysis.
• gammaCore reduced pain intensity over 3x greater than sham at 60 minutes and over 6x greater at 120 minutes
• gammaCore reduced the need for other rescue medications
Consider gammaCore: a novel approach to treatment for cluster headache
Cluster headache is a debilitating condition that is very hard to treat.
gammaCore is the first and only therapy FDA-cleared for the prevention of cluster headache. When added to standard of care (SoC), gammaCore significantly reduced the number of weekly cluster attacks for many patients.3,6,7
gammaCore is also FDA-cleared to acutely treat the pain associated with episodic cluster headache. In 2 pivotal studies, gammaCore demonstrated fast, reliable, and safe pain reduction.3,8,9
Largest study of chronic cluster headache prevention that showed significant treatment effects6
The PREVA study was a multicenter, open-label, randomized, controlled, parallel-group study of adult patients who had been diagnosed with chronic cluster headache for at least 1 year prior to study enrollment.6

The primary endpoint of the PREVA study was the reduction in mean number of cluster headache attacks per week.6

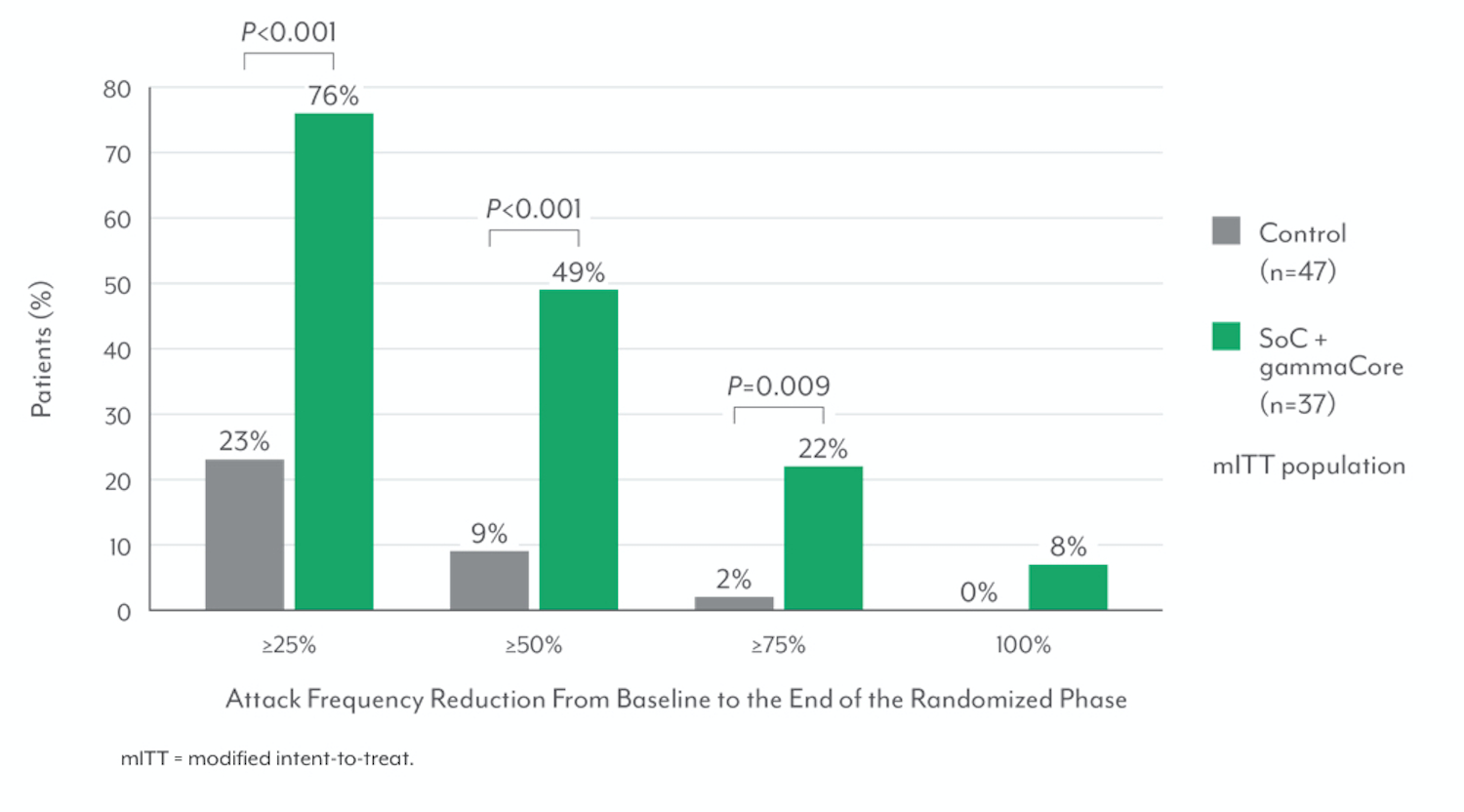
The secondary endpoints of the PREVA study were the ≥50% response rate (ie, proportion of participants with ≥50% reduction in mean number of cluster attacks per week) and acute medication use.6
Statistically significant reduction in weekly attacks and reliable response rates
Baseline vs the Last 2 Weeks of Randomized Phase6
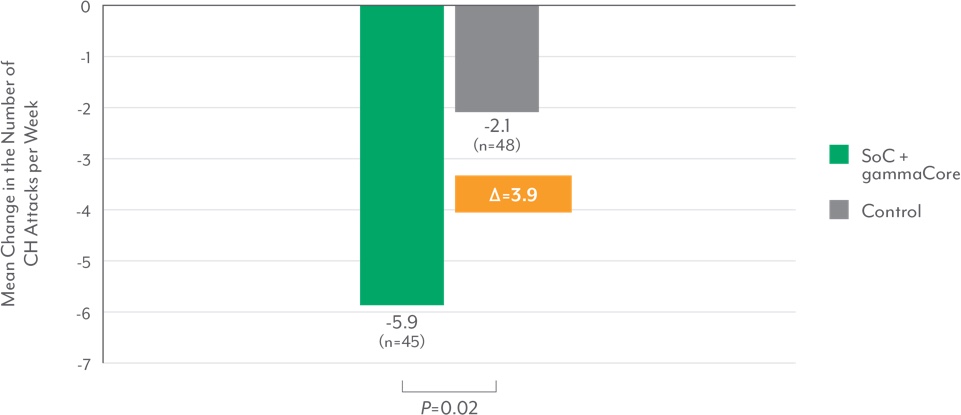
Attack frequency decreased 40% from baseline7
- Weekly attack frequency at the end of the randomized phase decreased by 40% from baseline with SoC + nVNS and increased by 1% with SoC alone, representing a 41% therapeutic benefit of nVNS (P<0.001)
Expanded Response Rates7
Decrease in use of acute treatments6
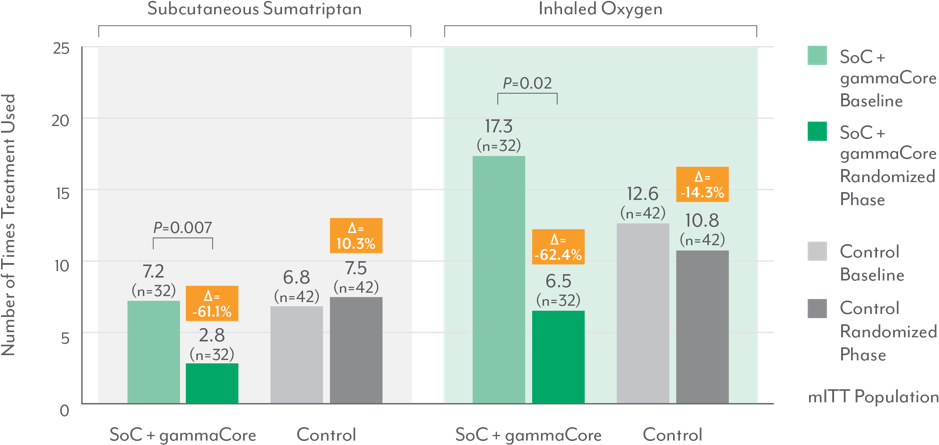
Significant reductions in the use of subcutaneous sumatriptan and inhaled oxygen
- During the randomized phase, the SoC + gammaCore (nVNS) group had a 57% decrease in the frequency of acute medication use (P<0.001)
One of the largest clinical programs conducted for cluster headache pain8

In the ACT1 Study, the primary endpoint was the percentage of people who reported “mild pain” or “no pain” 15 minutes after using gammaCore to treat their first attack.3,8

In the ACT2 Study, the primary endpoint was the percentage of total attacks that were pain-free 15 minutes after using gammaCore, with no use of rescue medication throughout the treatment period (30 minutes) and the option to repeat treatment approximately 3 minutes after the first treatment.3,9
Fast relief from the first attack3,8,9
gammaCore provided better relief at 15 minutes vs sham
Response in First Treated Attack (Primary Endpoint): All Study Groups3,8
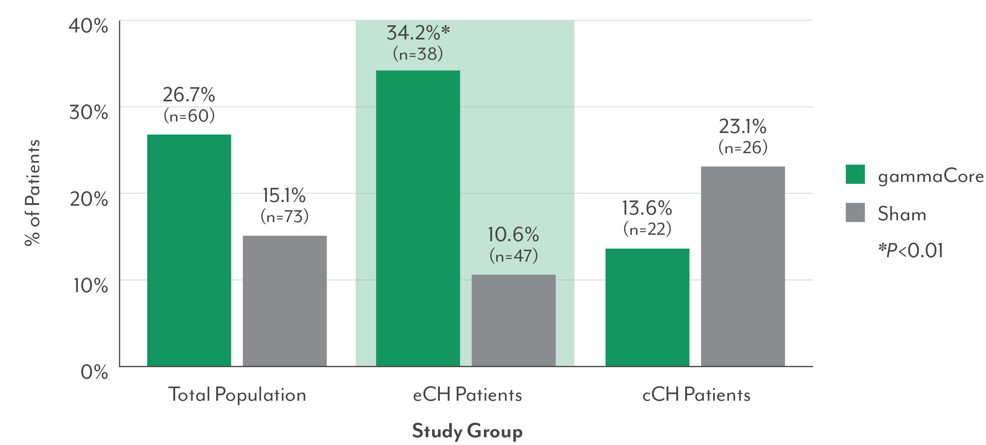

- Significantly more episodic cluster headache patients responded with gammaCore for the first treated cluster attack vs those receiving sham (response = reporting “mild pain” or “no pain” at 15 minutes)
Percentage of All Treated Attacks That Were Pain-Free at 15 Minutes (Primary Endpoint): All Study Groups3,9
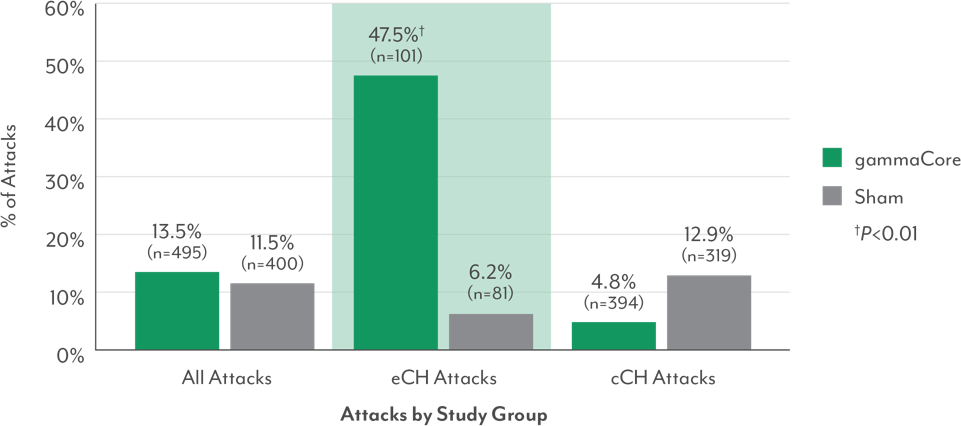

- Among episodic cluster headache patients, significantly more episodic cluster attacks treated with gammaCore were pain-free at 15 minutes vs those treated with sham
Reliable relief for at least half of treated attacks3,8,9
gammaCore provided reliable relief for more patients vs sham
Episodic Cluster Headache Patients Successfully Treated ≥50% of Attacks3,8‡
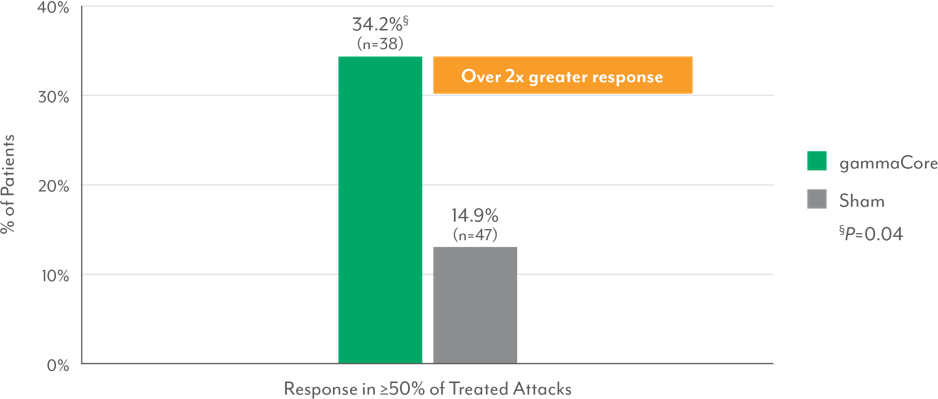
‡Response defined as "no pain" or "mild pain" at 15 minutes.

- Significantly more episodic cluster headache patients responded to gammaCore at 15 minutes for ≥50% of all treated attacks vs those receiving sham
Episodic Cluster Headache Patients Successfully Treated ≥50% of Attacks3,9‖
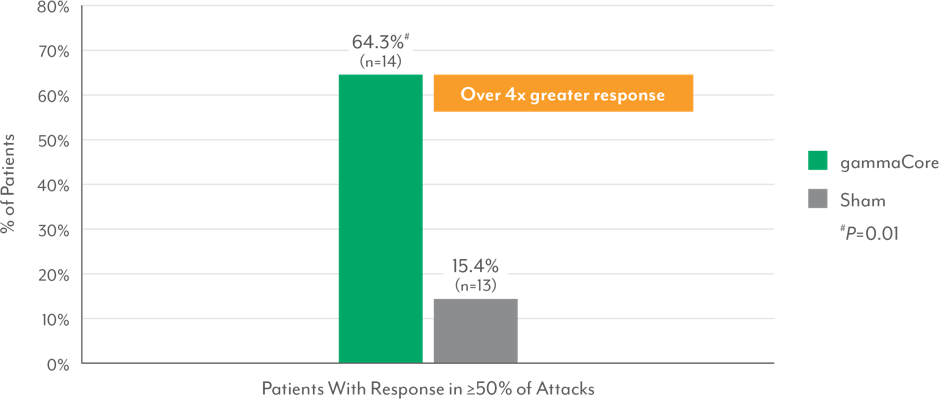
‖Response defined as “no pain” or “mild pain” at 15 minutes.

- Significantly more episodic cluster headache patients treated with gammaCore responded within 15 minutes in ≥50% of treated attacks vs those treated with sham
Reduction in the duration and intensity of attacks3,8
gammaCore decreased the length and the severity of attacks vs sham
Significant Reduction in Duration of First Treated Attack in Episodic Cluster Headache Patients3,7**
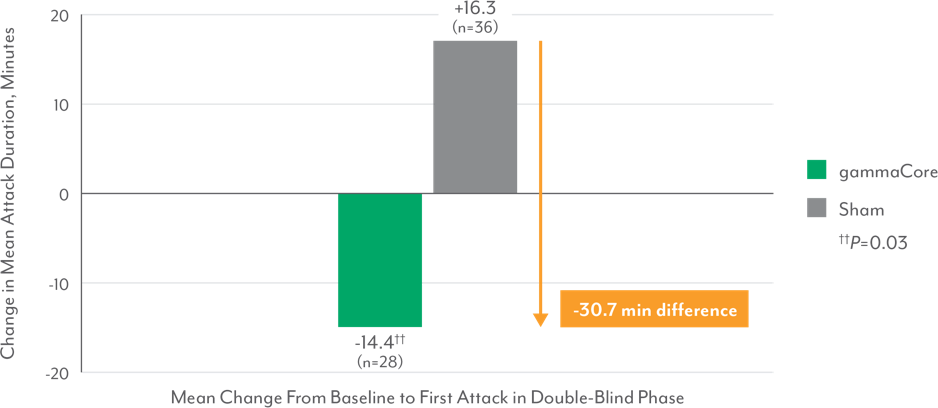
**Based on available patient data.

- Episodic cluster headache patients treated with gammaCore also had a significantly greater mean reduction in the duration of the treated attacks vs those treated with sham, who had an increase in attack duration
Significant Reduction in Pain Intensity Across All Treated Attacks in Episodic Cluster Headache Patients3,9‡‡
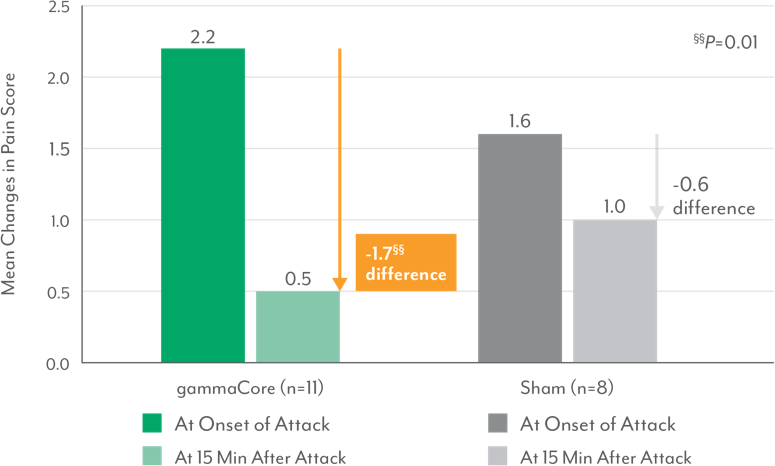
‡‡Mean of all attacks per patient.

- Episodic cluster headache patients treated with gammaCore also reported significantly greater reduction in pain intensity at 15 minutes vs those treated with sham
References: 1. Diener HC, Goadsby PJ, Ashina M, et al. Non-invasive vagus nerve stimulation (nVNS) for the preventive treatment of episodic migraine: the multicenter, double-blind, randomized, sham-controlled PREMIUM trial. Cephalalgia. 2019;39(12):1475–1487. 2. Silberstein SD, Calhoun AH, Lipton RB, et al. Chronic migraine headache prevention with noninvasive vagus nerve stimulation: the EVENT study. Neurology. 2016;87(5):529-5383. gammaCore Instructions for Use. Basking Ridge, NJ: electroCore, Inc.: 2020. 4. Data on file, electroCore, Inc. 5. Tassorelli C, Grazzi L, de Tommaso M, et al. Non-invasive vagus nerve stimulation as acute therapy for migraine: the randomized PRESTO study. Neurology. 2018;91(4):e364-e373. 6. Gaul C, Diener HC, Silver N, et al; PREVA Study Group. Non-invasive vagus nerve stimulation for PREVention and Acute treatment of chronic cluster headache (PREVA): a randomised controlled study. Cephalalgia. 2016;36(6):534-546. 7. Gaul C, Magis D, Liebler E, Straube A. Effects of non-invasive vagus nerve stimulation on attack frequency over time and expanded response rates in patients with chronic cluster headache: a post hoc analysis of the randomised, controlled PREVA study. J Headache Pain. 2017;18(1):22. 8. Silberstein SD, Mechtler LL, Kudrow DB, et al; ACT1 Study Group. Non-invasive vagus nerve stimulation for the acute treatment of cluster headache: findings from the randomized, double-blind, sham-controlled ACT1 study. Headache. 2016;56(8):1317-1332. 9. Goadsby PJ, de Coo IF, Silver N, et al; ACT2 Study Group. Non-invasive vagus nerve stimulation for the acute treatment of episodic and chronic cluster headache: a randomized, double-blind, sham-controlled ACT2 study. Cephalalgia. 2018;38(5):959-969.